- Product Listings
- Compare Products
- Calculators
- Product Tours
- Expert Reviews
- Our Experts
- Company Directory
- About Beye
- Contact Us
Ⓒ 2026 Beye.com. All rights reserved.
This content is intended for health care professionals and providers only. The information contained on Beye.com, including text, graphics, images, and interactive activities, is for informational purposes only, and is not intended to be a substitute for professional medical advice. Beye LLC, via its Editors and Publisher, accepts no responsibility for any injury or damage to persons or property occasioned through the implementation of any ideas or use of any product described herein. Although great care is taken to ensure that all information is accurate, it is recommended that readers seek independent verification of advice on drugs and other product usage, surgical techniques and clinical processes prior to their use. References made in article may indicate usage of medical equipment or drugs at dosages, for periods of time, and in combination not included in the current prescribing information. Inclusion of advertising materials on the website thereof, does not constitute and representation or guarantee by Beye LLC of the quality of such products, or of the claims made.
Compare Microinvasive Glaucoma Surgery (MIGS)
15 Products
reset all
At-a-Glance
Description
FDA
CE Mark
Classification
Indication
Mechanism of Action
Usage
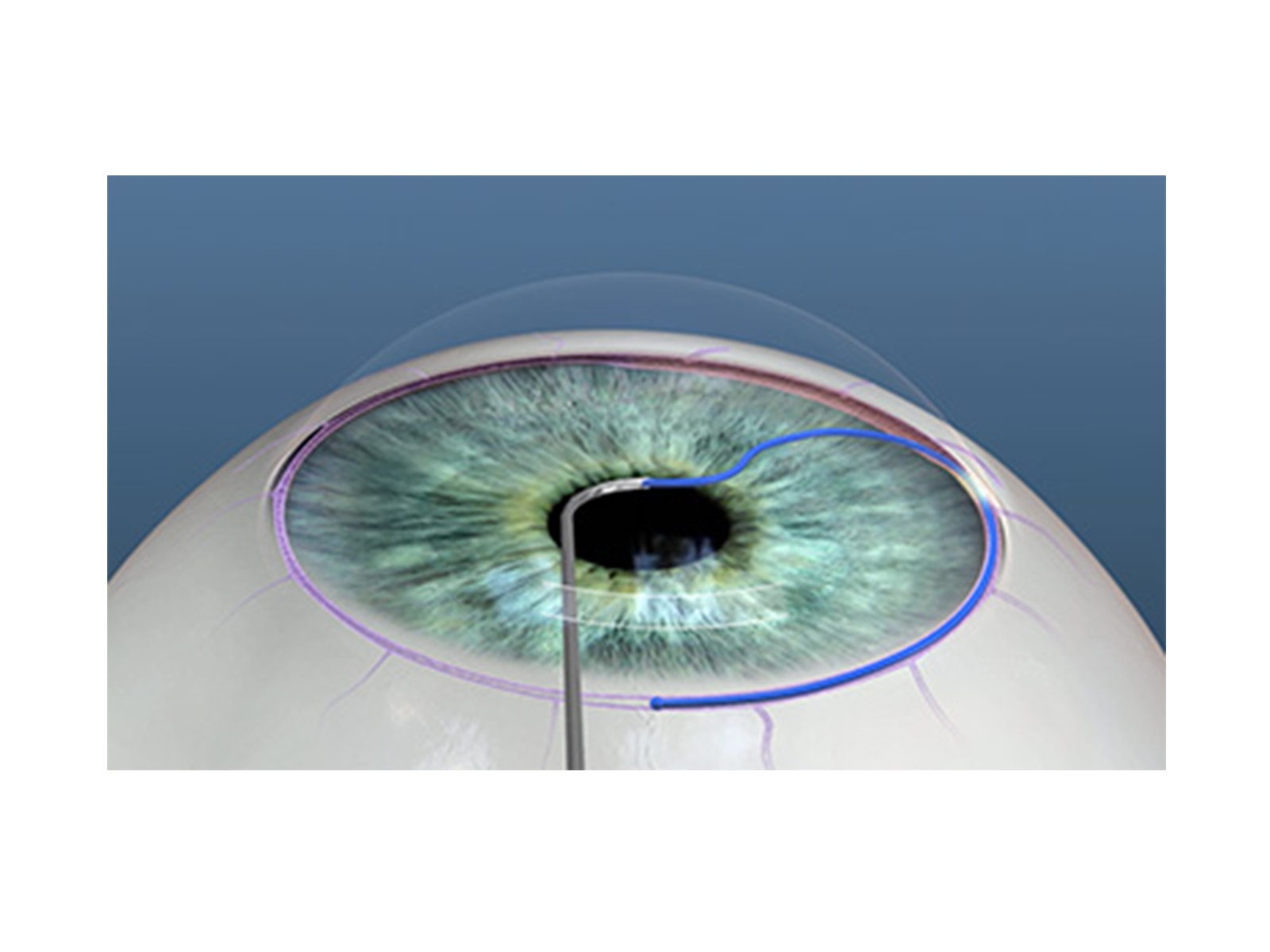
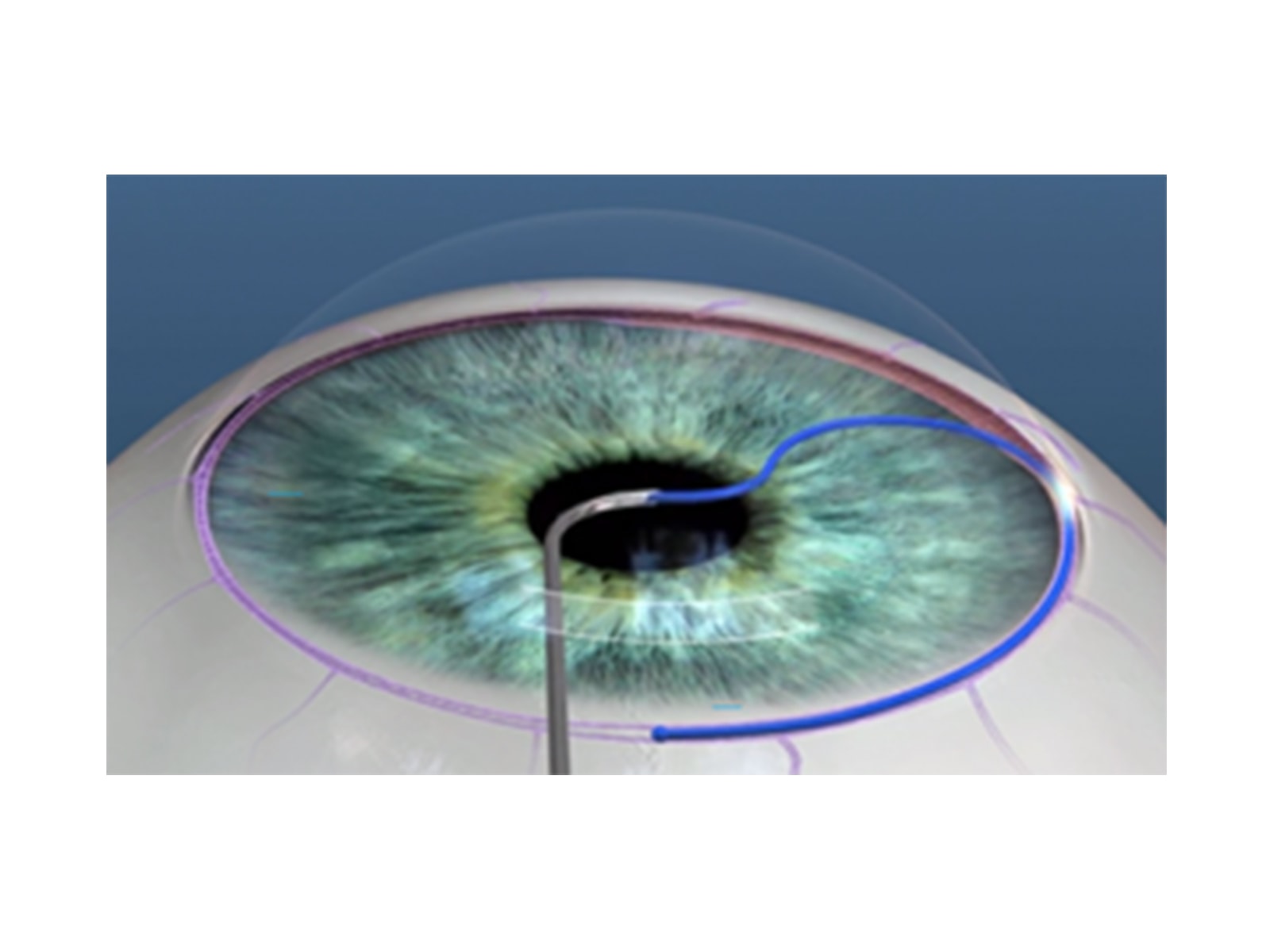
- Controlled entirely by the surgeon and can be used in viscodilation of Schlemm canal and any application of viscoelastic-assisted dissection within the anterior segment
- Easy-to-use roller wheel mechanism simultaneously retracts the microcatheter and triggers the internally integrated pump mechanism to deliver a predetermined amount of viscoelastic
- Can be used in a stand-alone procedure or in conjunction with cataract or other anterior segment surgeries
- The single-use instrument does not require a capital investment and is reimbursed under a Category I CPT code
A procedure based surgical intervention that facilitates an ab interno approach for controlled viscodilation of Schlemm canal and viscoelastic-assisted dissection within the anterior segment
Yes
Yes
Ab interno
For the delivery of small amounts of viscoelastic fluid such as Healon or HealonGV from Abbott Medical Optics (AMO), Amvisc from Bausch & Lomb, or PROVISC from Alcon during ophthalmic surgery.
Microcatheterization and viscodilation of Schlemm canal and collapsed collector channels
Treatment of mild to severe glaucoma during cataract surgery or as a stand-alone procedure and viscoelastic-assisted dissection techniques within the anterior segment
- Material: Polyimide (PI) polymer
- The device has an inner diameter of 300 microns, outer diameter of 430 microns, and overall length of 6.35 mm
- Three (3) retention rings prevent migration of device and serve as landmarks during placement
- 64 fenestrations ensure that the auxiliary orifices continue to filter the aqueous humor in case the distal end gets blocked
The CyPass Micro-Stent is made from a biocompatible material used in intraocular lens haptics and is designed to enhance outflow to the uveoscleral pathway via the supraciliary space by bypassing the ciliary body. The micro-stent is inserted via the Applier which has a self-locating, semi-blunt tip designed to help the stent slide in the virtual space between the ciliary body and the sclera without causing any adjacent tissue damage.
Voluntary Recall
Voluntary Recall
Ab interno
For use in conjunction with cataract surgery for the reduction of intraocular pressure (IOP) in adult patients with mild-to-moderate primary open-angle glaucoma (POAG).
Enhances outflow to the uveoscleral pathway via the supraciliary space by bypassing the ciliary body
Treatment of mild to moderate glaucoma at the time of cataract surgery
- Material: Surgical-grade stainless steel
- The device measures 2.64 mm and have a 27 guage shaft with a 0.4 mm outer diameter
- The spur and faceplate prevent extrusion and intrusion respectively
The EX-PRESS Glaucoma Filtration Device is implanted in an ab externo procedure under a half-thickness scleral flap, creating a subconjunctival filtration bleb. A beveled tip allows for precise, controlled insertion and relief port on the side of the lumen allows for continued drainage even in the event of blockage of the tip.
Yes
Yes
Ab externo
To reduce intraocular pressure in glaucoma patients where medical and conventional surgical treatments have failed.
Channels aqueous humor from the anterior chamber to a subconjunctival filtration bleb
Treatment of glaucoma in patients as indicated and can be performed during cataract surgery or as a stand-alone procedure
- Material: Gelatin, cross-linked with glutaraldehyde
- Injected through a 27-gauge, double-beveled needle
The XEN Gel Stent is a 6-mm gel stent designed to be implanted through a small corneal incision to create a new pathway for aqueous flow from the anterior chamber to the subconjunctival space. The stent, which swells slightly and becomes flexible when hydrated, creates a permanent channel for aqueous outflow and overtime creates a low-lying bleb.
Yes
Yes
Ab interno
For the management of refractory glaucomas, including cases where previous surgical treatment has failed, cases of primary open-angle glaucoma, and pseudoexfoliative or pigmentary glaucoma with open angles that are unresponsive to maximum tolerated medical therapy.
Not specified
Treatment of mild to severe glaucoma during cataract surgery or as a stand-alone procedure as indicated
- Material: Surgical-grade Nonferromagnetic Titanium with heparin coating
- The device measures 1 mm in length, 0.33 mm in height, and has a 120 micron lumen diameter
- Preservation of the trabecular meshwork ensures a natural episcleral back pressure of 8-11 mm Hg with minimal risk of hypotony
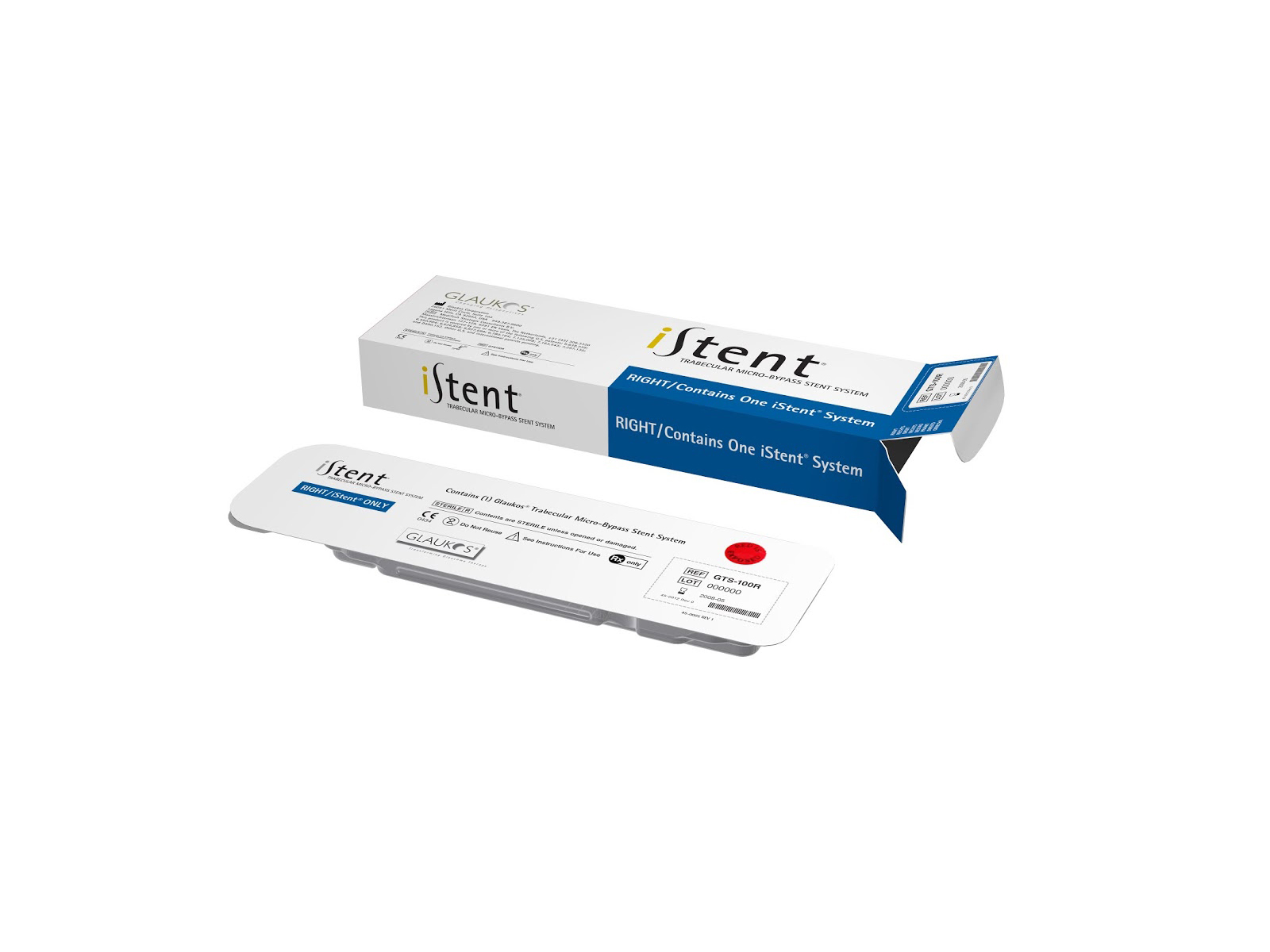
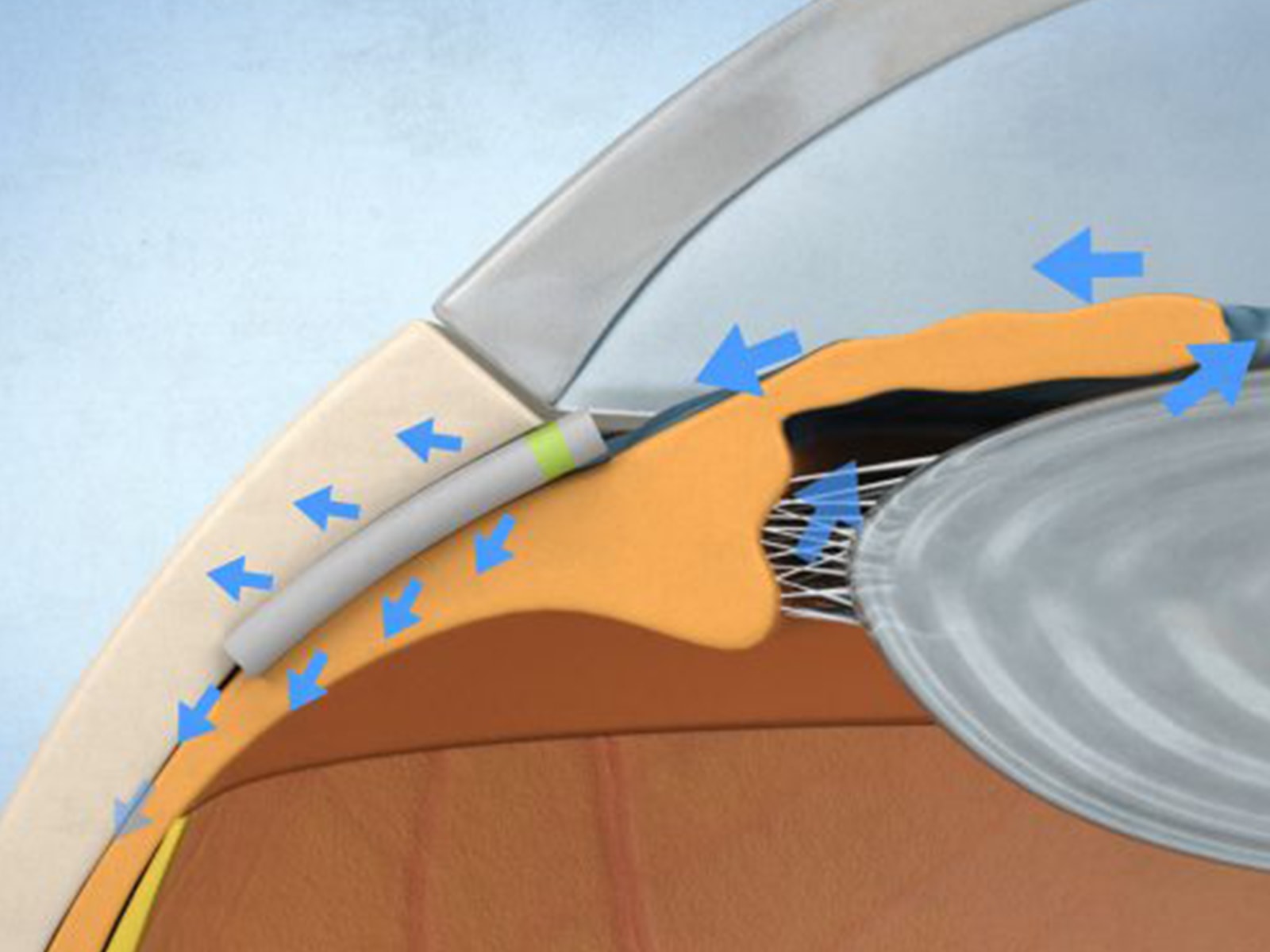
The iStent is engineered to fit into Schlemm canal, enabling steady physiologic outflow from a snorkel projecting into the anterior chamber. The self-trephinating tip guides device through the trabecular meshwork while retention arches secure placement.
Yes
Yes
Ab interno
For use in conjunction with cataract surgery for the reduction of IOP in adult patients with mild to moderate open-angle glaucoma currently treated with ocular hypotensive medication
Creates a channel through trabecular meshwork to enable aqueous humor to flow from the anterior chamber into the Schlemm’s canal
Treatment of mild to moderate glaucoma at the time of cataract surgery in patients as indicated
- Material: Polymer
- Ridged tube designed to improve implant retention in its desired location
The iStent Supra is designed to reduce IOP by accessing the suprachoroidal space in the eye. Approximately 4.0 mm in length and curved to follow the eye?s anatomy, the iStent Supra is already approved for marketing in the European Union and certain other countries outside the United States.
Yes
Yes
Ab interno
Not specified
Creates a channel to enable aqueous outflow to the suprachoroidal space
Treatment of mild to moderate glaucoma during cataract surgery or as a stand-alone procedure
- Material: Surgical-grade Nonferromagnetic Titanium with heparin coating
- Each stent measures 0.36 mm in height and 0.23 mm in width (about one-third the size of the first-generation iStent)
- Preservation of the trabecular meshwork ensures a natural episcleral back pressure of 8-11 mm Hg with minimal risk of hypotony
- Prior to premarket approval (PMA) from the FDA, more than 30,000 iStent injects had been implanted in the European Union, Armenia, Australia, Brazil, Canada, Hong Kong, Singapore and South Africa
The iStent inject Trabecular Micro-Bypass System entails an injector system preloaded with two micro-scale stents that the surgeon places sequentially through a single corneal entry point directly into two locations within the trabecular meshwork to restore the natural, physiological outflow.
Yes
Yes
Ab interno
For the reduction of IOP in adult patients with mild-to-moderate primary open-angle glaucoma currently treated with ocular hypotensive medication
Enable aqueous outflow from the anterior chamber into Schlemm canal
Treatment of mild to moderate glaucoma during cataract surgery
- Material: STAR (medical-grade silicone)
- Material is biocompatible, non-degradable substance developed at the University of Washington in Seattle
- The MINIject first-in-man clinical trial is currently undergoing regulatory approval
The MINIject implant is made from a proprietary material with a unique structure composed of an organized network of hollow spheres which encourage a natural flow speed. According to the company, the porous design reduces the incidence of fibrosis and minimises scarring, thus increasing implant durability.
No
No
Ab interno
Not specified
Enhancing aqueous humour outflow from the anterior chamber to the suprachoroidal space
Not specified
- Material: Nitinol and titanium oxide coating
- Unique design allows aqueous flow through both the inner lumen and frames of the scaffold
- Ensures dilation along 90 degrees of Schlemm canal
Hydrus Microstent is designed to be inserted into Schlemm canal to enable circumferential outflow and function like a scaffold to prevent collapse of the canal. The device is made from nitinol, a super-elastic, biocompatible alloy.
Yes
Not specified
Ab interno
Not specified
Functions like a scaffold to prevent collapse of the canal and enables circumferential outflow of aqueous humor through lumen openings
Treatment of mild to moderate glaucoma at the time of cataract surgery in patients currently on hypotensive medication
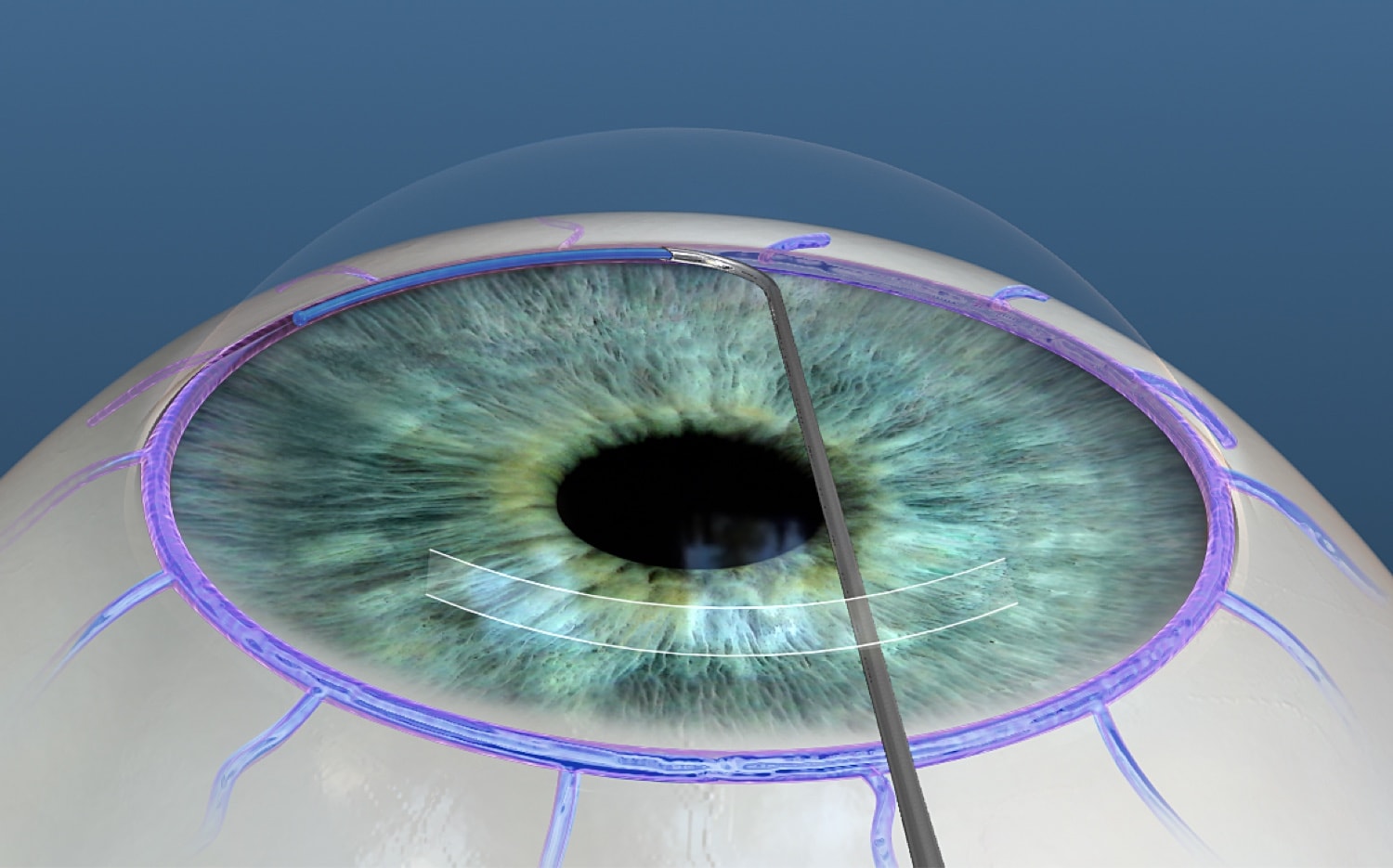
- Enables removal of the nasal 60 to 120 degrees of the trabecular meshwork and the inner wall of Schlemm canal via microcautery
- Continuous irrigation & aspiration removes debris and regulates temperature
- Procedure does not rely on foreign body inside the eye
The Trabectome is an ab interno technique that involves the removal of the nasal 60 to 120 degrees of the trabecular meshwork and the inner wall of Schlemm canal via bipolar electrocautery. The system consists of a handpiece, which has built-in continuous irrigation, a power console, and a 3-stage foot pedal control.
Yes
Yes
Ab interno
Not specified
Ablates the trabecular meshwork and inner wall of Schlemm canal
Not specified
- Dual blades positioned for precise parallel incisions of the trabecular meshwork with minimal residual leaflets
- Procedure does not rely on foreign body inside the eye
- Does not require a capital outlay; the cost is limited to that of the blade
The Kahook Dual Blade (KBD) is a single-use, surgical grade stainless steel instrument consisting of a long, thin shaft that allows for access across the anterior chamber, a pointed tip used to pierce trabecular meshwork, a ramp that lifts and stretches the tissue as the device is advanced, and two parallel blades that cut a strip of tissue.
Yes
Yes
Ab interno
Not specified
Removes a section of trabecular tissue to provide aqueous direct access to the collector channels and the distal outflow system
Treatment of mild to severe glaucoma during cataract surgery or as a stand-alone procedure
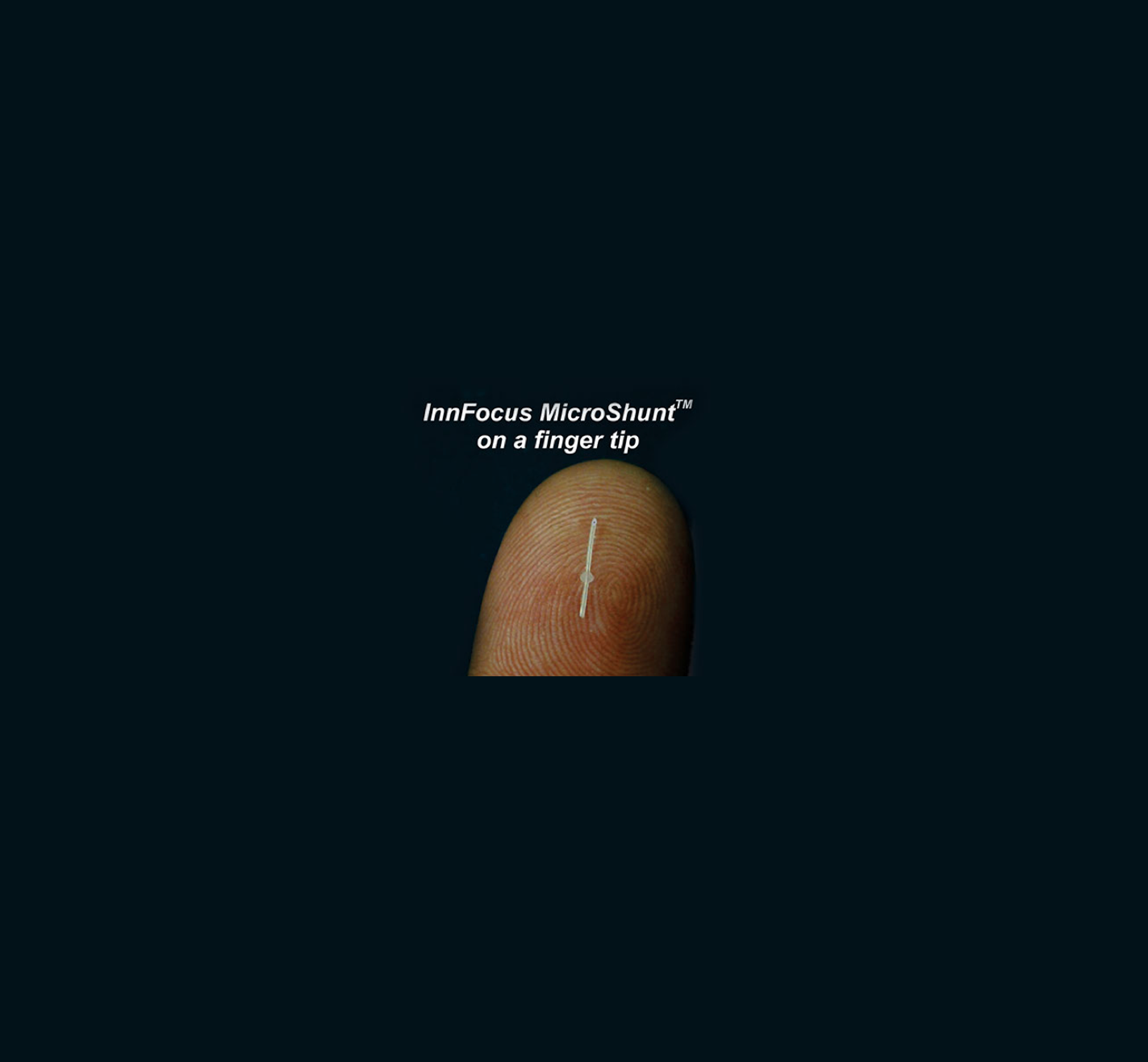
- Material: SIBS (Poly(Styrene-block-IsoButylene-block-Styrene))
- Currently is the final phase of clinical study in the world's only randomized trial against trabculectomy
The InnFocus MicroShunt Glaucoma Drainage System (formerly known as MIDI Arrow) was developed in conjunction with the University of Miami's Bascom Palmer Eye Institute. The drainage implant consists of a small micro-tube made of a biostable thermoplastic elastomeric material that shunts aqueous fluid from the anterior chamber of the eye to a sub-conjunctival/sub-Tenon flap. The shunt was designed to be thin and soft to conform to the curvature of the eye.
No
Yes
Ab externo
Ab externo with mitomycin C
Shunts fluid from anterior chamber to subconjunctival space under a subconjunctival sub-Tenon’s flap.
Treatment of mild to severe open-angle glaucoma during cataract surgery or as a stand-alone procedure
- Introduced through a single, clear corneal incision and easy-to-use roller wheel mechanism advances the atraumatic polymer tip through Schlemm canal
- Can be used in a stand-alone procedure or in conjunction with cataract surgery
- The single-use instrument does not require a capital investment and is reimbursed under a Category I CPT code
- Procedure does not rely on foreign body inside the eye
The TRAB360 device is a manual trabeculotome instrument capable of cutting up to all 360 degrees of trabecular meshwork via an ab interno approach. A blunt polymer trabeculotome is advanced through the tip of a curved cannula with a roller gear mechanism and mechanically cleave trabecular meshwork when it is withdrawn from the eye.
Yes
Yes
Ab interno
For the manual cutting of the trabecular meshwork, or trabeculotomy. TRAB360 can be used to mechanically cut up to 360 degrees of trabecular meshwork.
Trabecular Meshwork and inner wall of Schlemm Canal
Treatment of mild to severe glaucoma during cataract surgery or as a stand-alone procedure
- Introduced through a single, clear corneal incision and easy-to-use roller wheel mechanism advances the atraumatic polymer tip through Schlemm canal and simultaneously delivers a controlled amount viscoelastic fluid to dilate the canal and collector channels
- Can be used in a stand-alone procedure or in conjunction with cataract surgery
- The single-use instrument does not require a capital investment and is reimbursed under a Category I CPT code
- Procedure does not rely on foreign body inside the eye
The OMNI Combined Procedure System viscodilates up to 360? of Schlemm canal and unroofs up to 360? of the trabecular meshwork. A blunt, polymer microcatheter is advanced through the tip of a curved cannula with a roller gear mechanism and delivers a controlled amount of viscoelastic fluid as it is retracted. After completing viscodilation, the microcatheter can be withdrawn from the eye (without retracting it into the cannula) to mechanically cleave trabecular meshwork.
Yes
Yes
Ab interno
For the catheterization and transluminal viscodilation of Schlemm’s canal and the cutting of trabecular meshwork to reduce intraocular pressure in adult patients with open-angle glaucoma.
Trabecular Meshwork and inner wall of Schlemm Canal
Treatment of mild to severe glaucoma during cataract surgery or as a stand-alone procedure
- Material: pure biocompatible gold
- Ab externo, with or without cataract extraction
Shunts aqueous from the anterior chamber to the suprachoroidal space.
No
No
Ab externo
Not specified
Enable aqueous outflow from anterior chamber to suprachoroidal space
Can be performed during cataract surgery or as a stand-alone procedure
-
HFDS can be used individually and can also be ideally combined with cataract surgery. HFDS is characterised by a very short intervention time. Bleb formation is prevented, and so is fibroblast movement to the sclerotomy. Moreover, there are no corneal scars. Oertli’s High Frequency Deep Sclerotomy boasts excellent long-term results. If required, a further HFDS intervention can be made to optimise results
HFDS stands for High Frequency Deep Sclerotomy. In micro-invasive glaucoma surgery (MIGS), HFDS creates direct access from the anterior chamber to the Schlemm’s canal and further into the sclera. The resistance of the trabecular meshwork is thus bypassed. The HFDS tip is inserted through 1.2 mm paracentesis and, by means of diathermy output, it creates six small sclerotomy pockets in the iridocorneal angle, ensuring improved outflow of aqueous fluid.
No
Yes
ab Interno
For use in primary open-angle glaucoma (POAG) and is intended to be used for bipolar diathermal ab-interno deep sclerotomy
Creates direct access to the Schlemm’s canal from the anterior chamber, thus avoiding outflow resistance of the trabecular meshwork
Alone or combined with cataract surgery