- Product Listings
- Compare Products
- Calculators
- Product Tours
- Expert Reviews
- Our Experts
- Company Directory
- About Beye
- Contact Us
Ⓒ 2026 Beye.com. All rights reserved.
This content is intended for health care professionals and providers only. The information contained on Beye.com, including text, graphics, images, and interactive activities, is for informational purposes only, and is not intended to be a substitute for professional medical advice. Beye LLC, via its Editors and Publisher, accepts no responsibility for any injury or damage to persons or property occasioned through the implementation of any ideas or use of any product described herein. Although great care is taken to ensure that all information is accurate, it is recommended that readers seek independent verification of advice on drugs and other product usage, surgical techniques and clinical processes prior to their use. References made in article may indicate usage of medical equipment or drugs at dosages, for periods of time, and in combination not included in the current prescribing information. Inclusion of advertising materials on the website thereof, does not constitute and representation or guarantee by Beye LLC of the quality of such products, or of the claims made.
Laser Systems
Sub-Category
Company
+ Show more
Filter By
Filter By
FDA
CE Mark
Pulse
Source
Spot size
Wavelength
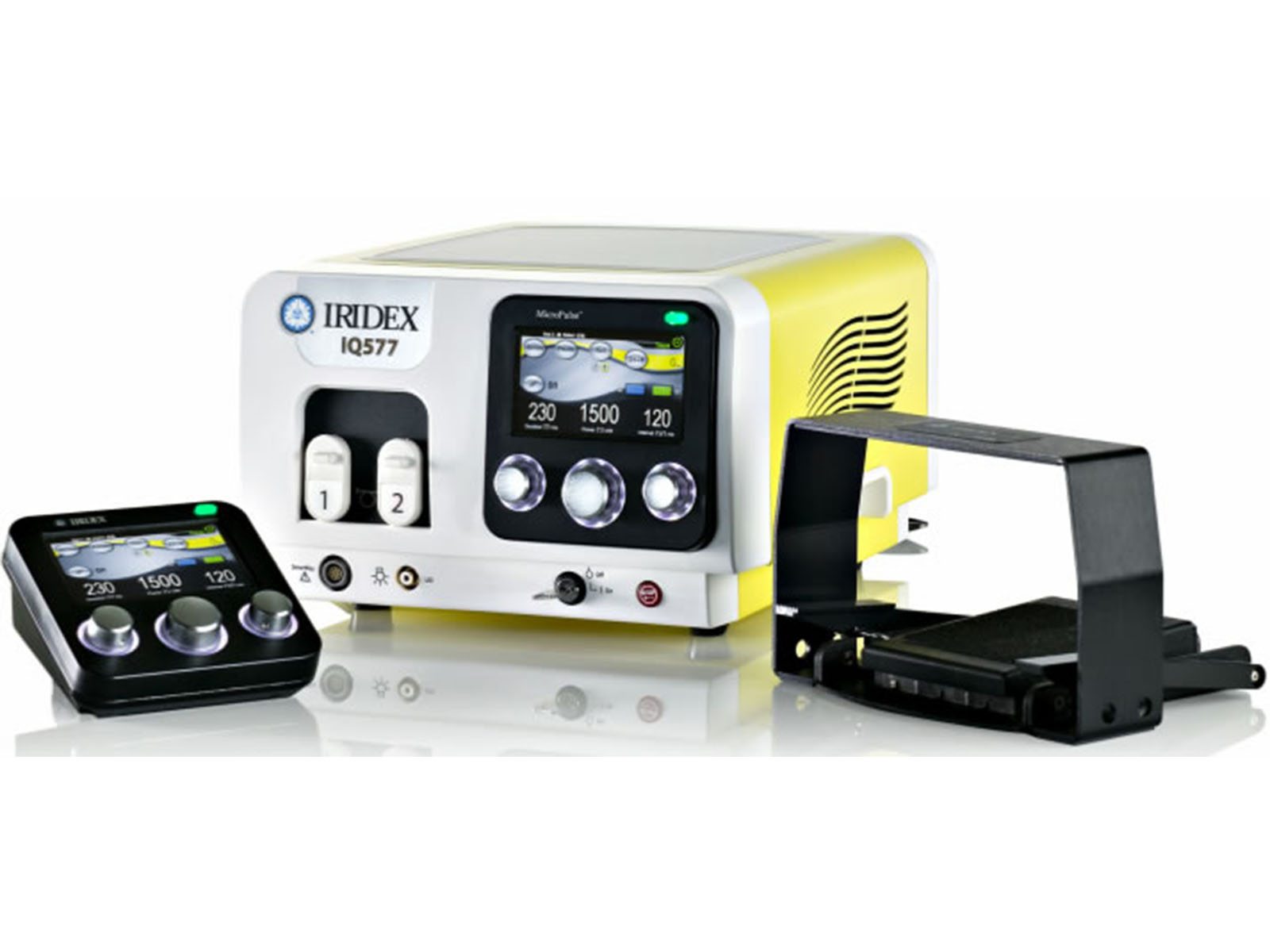
IQ 577 Laser System
Iridex Corporation
E2 Microprobe Laser and Endoscopy System
Beaver-Visitec International
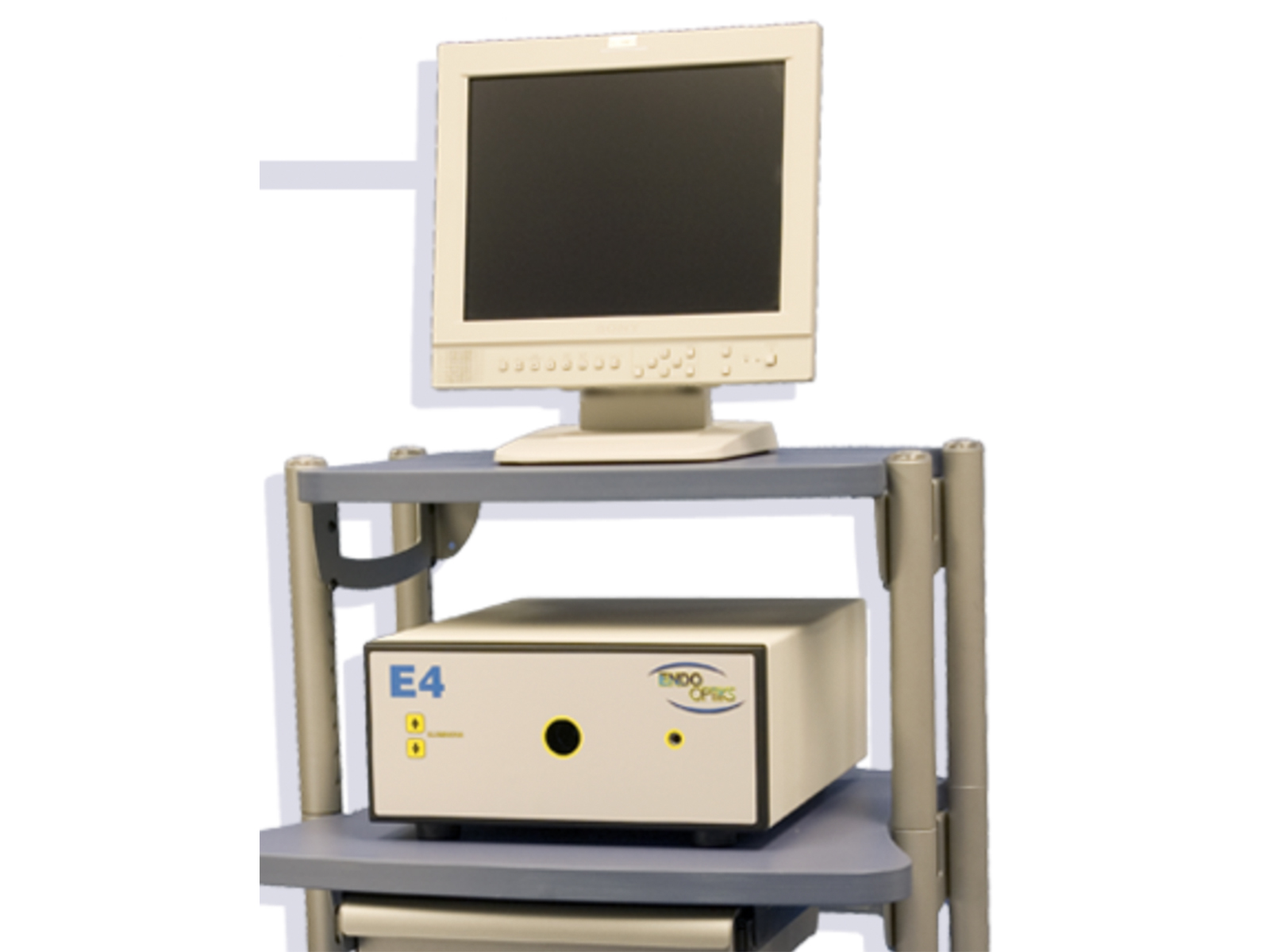
E4 Ophthalmic Endoscopy System
Beaver-Visitec International
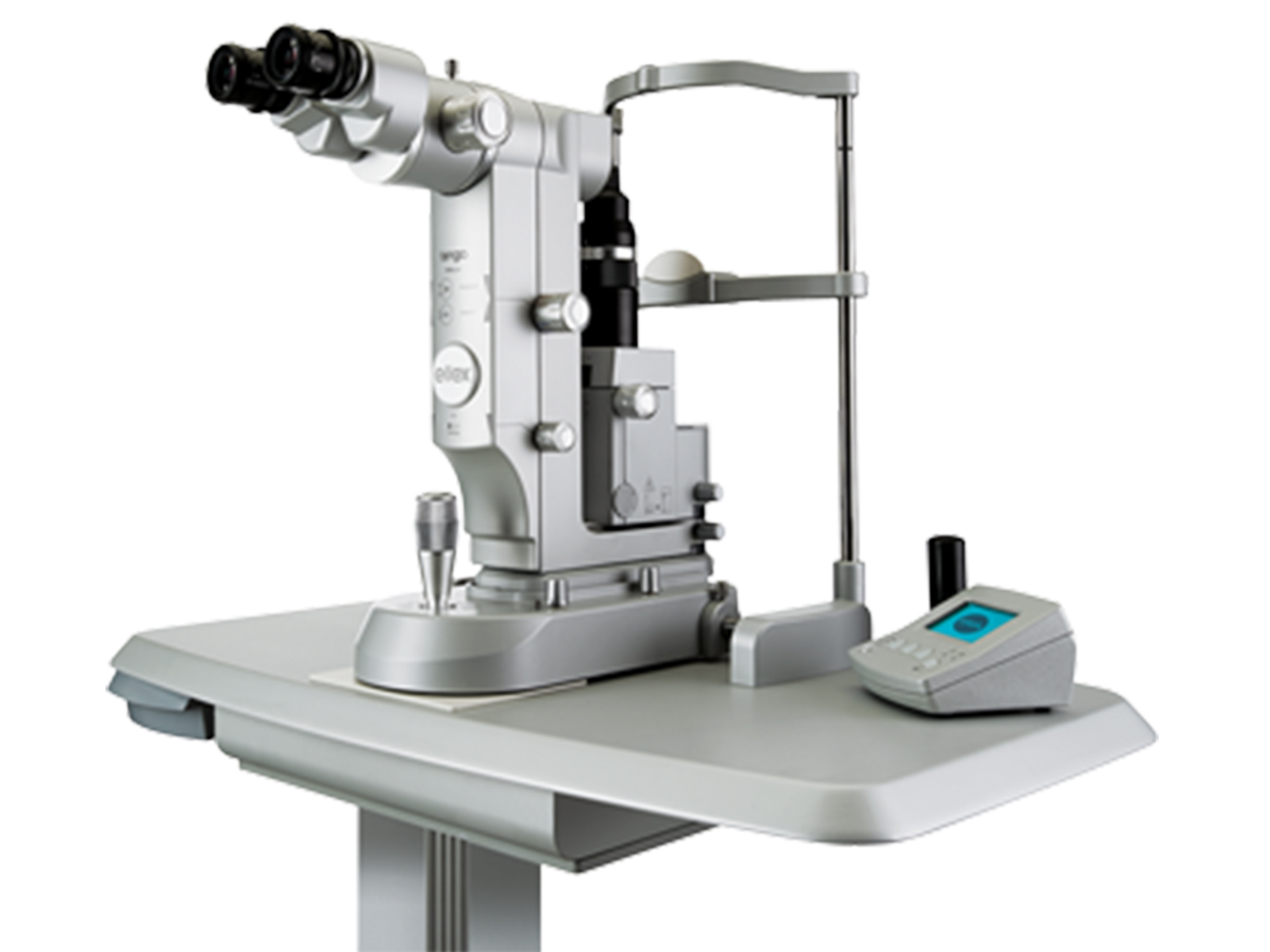
VISULAS YAG III
Carl Zeiss Meditec, Inc.
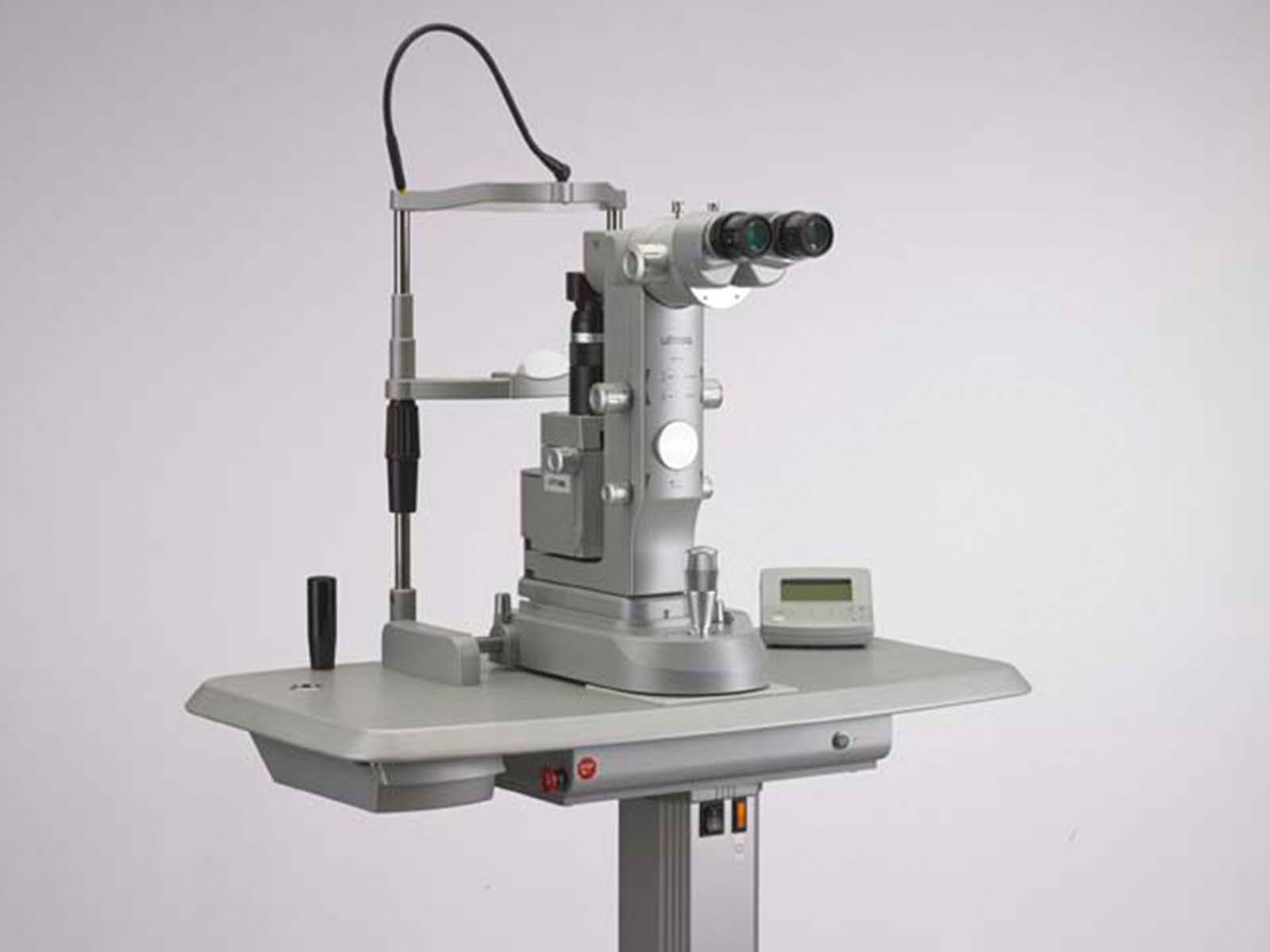
Solo
Ellex Inc.
Ultra Q Reflex
Ellex Inc.
Tango Reflex
Ellex Inc.
G-Probe Glaucoma Device
Iridex Corporation
Cyclo G6 Glaucoma Laser System
Iridex Corporation
Optimis Fusion
Quantel Medical
LIGHTLas SLT
LightMed Corporation
LIGHTLas YAG
LightMed Corporation
Selecta II
Lumenis
YC-1800
Nidek, Inc.
SoLuTis
Quantel Medical
Optimis II
Quantel Medical
IQ 810 Laser System
Iridex Corporation
Selecta Duet
Lumenis
LIGHTLas YAG/SLT Deux
LightMed Corporation
+ Show More